Whoa, thank you for joining us to learn more about the national bioengineered food disclosure standard. As many of you are aware, in the summer of 2016, Congress passed an amendment to the Agricultural Marketing Act of 1946. That amendment required the Secretary of Agriculture to draft regulations to establish a national mandatory bioengineered food disclosure standard. A team of dedicated individuals from the Agricultural Marketing Service and other agencies across the USDA have worked diligently over the last two years to draft the final regulations. By providing a uniform national standard for labeling bioengineered foods, we can increase transparency in our food system and give consumers information about the bioengineered status of their foods. While doing so, we avoid a patchwork of state labeling regulations that could be confusing for consumers and expensive for manufacturers. To get to this point, the team at USDA relied heavily on information and input from stakeholders across the landscape, including producers, food processors, food manufacturers, retailers, and consumers. This included over 112,000 responses to questions AMS posed on its website and more than 14,000 comments after AMS published the proposed rule earlier this year. Thank you to everybody who provided input. It has been invaluable in designing a standard that provides consumers with the information they are looking for while giving those subject to the standard necessary guidance and, where appropriate, added flexibility. This presentation is just one of many different steps USDA and AMS are taking to help those regulated by the standard understand and implement the final regulations. Along with this recording, we've put fact sheets and frequently asked questions on the website, which we'll continue to update as we receive questions from all of you. While we've been limited by the rulemaking process in how much we've been able to communicate with external stakeholders...
Award-winning PDF software





2018 allowable living expenses national standards Form: What You Should Know
IRS National Standards for Allowable Living Expenses May 1, 2025 — National Standards have been established for four necessary expenses: clothing, travel and incidental, shelter and meal expenses IRS National Standards for Allowable Living Expenses May 1, 2025 — Cases Filed Between May 1, 2018, and December 31, 2018, Inclusive; 333 · 31 · 84 · 39 ; 606 · 56 · 115 · 70 Means-Testing-2018-May.pdf The information provided below is applicable to Official Bankruptcy Forms Bankruptcy Allowable Living Expenses — National Standards (See 11 U.S.C. § 1053(d)). IRS National Standards for Allowable Living Expenses July 1, 2025 — National Standards have been established for three necessary expenses: housing, utilities and meals IRS National Standards for Allowable Living Expenses June 18, 2025 — National Standards have been established for two necessary expenses: housing, utilities and meals IRS National Standards for Allowable Living Expenses June 18, 2025 — Cases Filed Between June 18, 2018, and December 31, 2018, Inclusive; 317 · 29 · 74 · 36 ; 634 · 51 · 123 · 64 Means-Testing-2018-July.pdf The information provided below is applicable to Official Bankruptcy Forms Bankruptcy Allowable Living Expenses — National Standards (See 11 U.S.C.
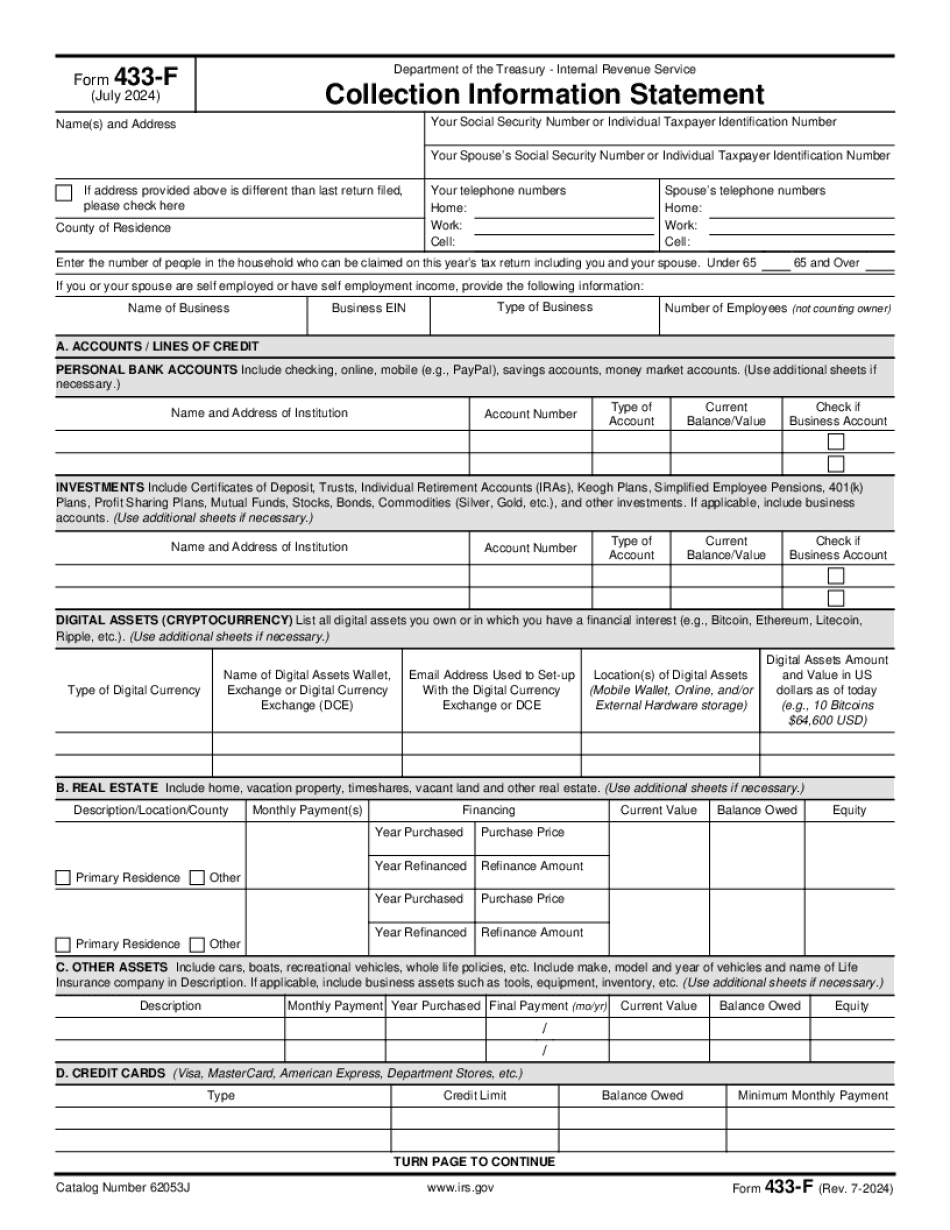
online solutions help you to manage your record administration along with raise the efficiency of the workflows. Stick to the fast guide to do Form 433-F, steer clear of blunders along with furnish it in a timely manner:
How to complete any Form 433-F online: - On the site with all the document, click on Begin immediately along with complete for the editor.
- Use your indications to submit established track record areas.
- Add your own info and speak to data.
- Make sure that you enter correct details and numbers throughout suitable areas.
- Very carefully confirm the content of the form as well as grammar along with punctuational.
- Navigate to Support area when you have questions or perhaps handle our assistance team.
- Place an electronic digital unique in your Form 433-F by using Sign Device.
- After the form is fully gone, media Completed.
- Deliver the particular prepared document by way of electronic mail or facsimile, art print it out or perhaps reduce the gadget.
PDF editor permits you to help make changes to your Form 433-F from the internet connected gadget, personalize it based on your requirements, indicator this in electronic format and also disperse differently.
Video instructions and help with filling out and completing 2025 allowable living expenses national standards